Differences between Norco- and Vicodin
Contents
Norco vs. Vicodin[edit]
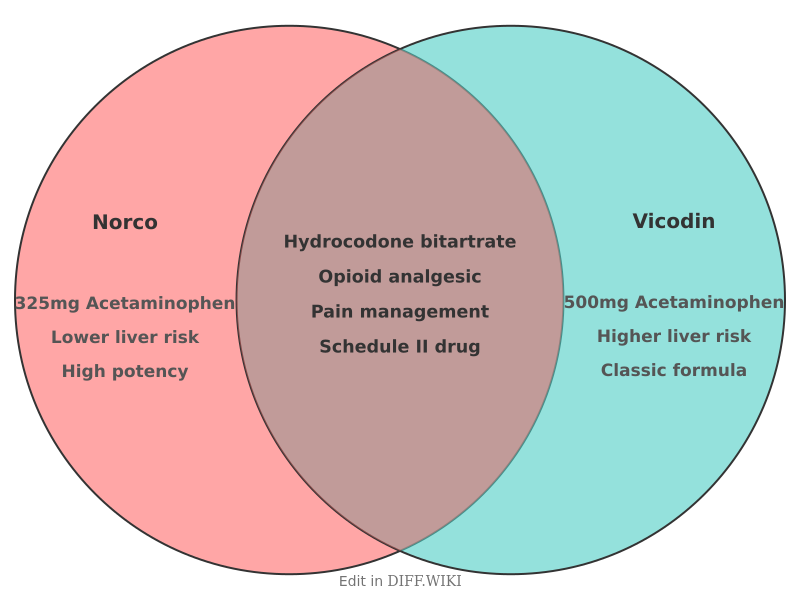
Norco and Vicodin are brand names for a fixed-dose combination drug consisting of hydrocodone bitartrate and acetaminophen. These medications are classified as opioid analgesics and are prescribed for the management of pain severe enough to require an opioid when alternative treatments are inadequate. Hydrocodone functions as an opioid agonist, while acetaminophen serves as a non-opioid analgesic and antipyretic.
The primary difference between these products historically involved the ratio of acetaminophen to hydrocodone. Prior to 2011, Vicodin was commonly distributed with 500 mg of acetaminophen per tablet, whereas Norco was formulated with 325 mg. Following a 2011 U.S. Food and Drug Administration (FDA) mandate, manufacturers were required to limit the amount of acetaminophen in prescription combination products to no more than 325 mg per dosage unit to reduce the risk of acetaminophen-induced liver injury.[1]
Comparison Table[edit]
| Category | Norco | Vicodin |
|---|---|---|
| **Active Ingredients** | Hydrocodone / Acetaminophen | Hydrocodone / Acetaminophen |
| **Hydrocodone Strengths** | 5 mg, 7.5 mg, 10 mg | 5 mg (Vicodin), 7.5 mg (ES), 10 mg (HP) |
| **Acetaminophen Strength** | 325 mg (standard) | 300 mg (current standard) |
| **DEA Schedule** | Schedule II | Schedule II |
| **Common Side Effects** | Constipation, nausea, somnolence | Constipation, nausea, somnolence |
| **Original Manufacturer** | Watson Pharmaceuticals | Knoll Pharmaceutical Company |
| **Primary Indication** | Moderate to severe pain | Moderate to severe pain |
| **Formulation Type** | Immediate-release tablet | Immediate-release tablet |
Regulatory and Safety History[edit]
In 2014, the Drug Enforcement Administration (DEA) moved hydrocodone combination products from Schedule III to Schedule II of the Controlled Substances Act. This reclassification followed recommendations from the FDA and the Department of Health and Human Services due to high rates of abuse and diversion associated with these medications.[2] Schedule II drugs are subject to stricter prescribing regulations, including a prohibition on refills; a new prescription is required for every dispending.
The reduction of acetaminophen in these medications was prompted by data linking high doses of the ingredient to acute liver failure. The FDA determined that limiting the acetaminophen content to 325 mg per tablet provided a safer margin for patients who might inadvertently take multiple acetaminophen-containing products simultaneously.[3] While Norco already utilized the 325 mg dose, Vicodin formulations were adjusted from their original 500 mg (Vicodin) and 750 mg (Vicodin ES) levels to 300 mg of acetaminophen to comply with safety guidelines.
Pharmacology and Administration[edit]
The hydrocodone component binds to mu-opioid receptors in the central nervous system, altering the perception of and response to pain. Acetaminophen is thought to inhibit prostaglandin synthesis in the central nervous system, though its exact mechanism is less understood than that of NSAIDs.
Both medications are administered orally. Dosage is adjusted according to the severity of the pain and the response of the patient. Because of the risk of respiratory depression and addiction, medical guidelines recommend using the lowest effective dose for the shortest duration possible.