Differences between Methadone and Suboxone
Contents
Methadone vs. Suboxone[edit]
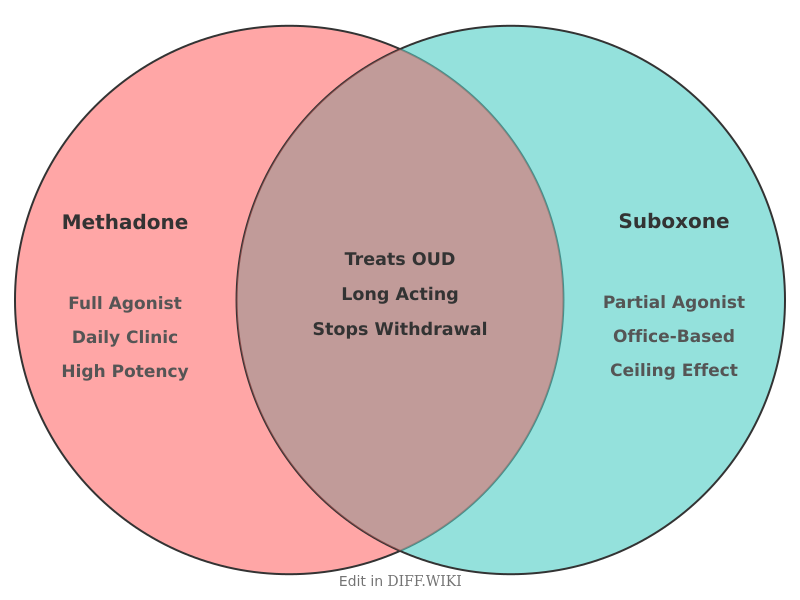
Methadone and Suboxone (a combination of buprenorphine and naloxone) are medications approved by the U.S. Food and Drug Administration (FDA) for the treatment of opioid use disorder (OUD).[1] While both drugs aim to reduce withdrawal symptoms and cravings, they possess distinct pharmacological profiles, regulatory requirements, and safety considerations.[2] Methadone is a long-acting full opioid agonist, while Suboxone is a partial opioid agonist and antagonist combination.[3][4]
Comparison Table[edit]
| Feature | Methadone | Suboxone (Buprenorphine/Naloxone) |
|---|---|---|
| Drug Class | Full opioid agonist | Partial opioid agonist / Antagonist |
| DEA Schedule | Schedule II | Schedule III |
| Induction | Can be started immediately | Requires mild-to-moderate withdrawal |
| Ceiling Effect | No; risk increases with dose | Yes; plateaus after certain doses |
| Dispensing Site | Certified Opioid Treatment Programs (OTPs) | Community pharmacies |
| Prescriber Requirements | Federal OTP certification | Any DEA-licensed practitioner |
| Cardiac Risk | Risk of QTc prolongation | Low to no risk reported |
| Half-life | 8–59 hours | 24–42 hours |
Pharmacological Mechanisms[edit]
Methadone activates the mu-opioid receptors in the brain to the same extent as other full agonists like heroin or oxycodone, but it does so more slowly and for a longer duration. This[4] provides stabilization without the rapid onset of euphoria when taken as prescribed. Because it has no "ceiling effect," higher doses continue to increase the suppression of respiratory function, which contributes to a higher risk of fatal overdose if misused.[5]
Suboxone consists of buprenorphine and naloxone in a 4:1 ratio. Buprenorphine is a partial agonist, meaning it only partially activates opioid receptors. After[6][7] a certain dose, its effects plateau, a phenomenon known as the ceiling effect. This characteristic reduces the risk of respiratory depression compared to full agonists. Naloxone is included to deter intravenous misuse; it has poor oral bioavailability but triggers immediate withdrawal if the medication is crushed and injected.[8]
Regulatory and Access Differences[edit]
Federal regulations in the United States mandate that methadone for OUD treatment be dispensed through certified Opioid Treatment Programs (OTPs). Patients typically visit these clinics daily for supervised dosing, although they may earn "take-home" privileges over time. In contrast, Suboxone can be prescribed by any practitioner with a standard DEA license following the removal of the "X-waiver" requirement in 2023. This allows patients to fill prescriptions at local pharmacies and manage their treatment in a primary care setting.[9]
[4]= Clinical Considerations =[edit]
Initiating Suboxone requires the patient to be in a state of opioid withdrawal. If buprenorphine is administered while full agonists are still active on the receptors, its high binding affinity will displace those agonists, causing "precipitated withdrawal." Methadone induction does not carry this risk and can be started while other opioids are still in the system. Methadone is often associated with QTc prolongation on an electrocardiogram (ECG), which may require cardiac monitoring in some patients, whereas buprenorphine is generally considered safer for those with cardiac concerns.[10]
References[edit]
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedref1 - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedref2 - ↑ Template:Cite web
- ↑ 4.0 4.1 4.2 Cite error: Invalid
<ref>tag; no text was provided for refs namedref3 - ↑ Template:Cite web
- ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedref4 - ↑ Cite error: Invalid
<ref>tag; no text was provided for refs namedref5 - ↑ Template:Cite web
- ↑ Template:Cite web
- ↑ Template:Cite journal