Differences between Celexa and Lexapro
Contents
Citalopram vs. escitalopram[edit]
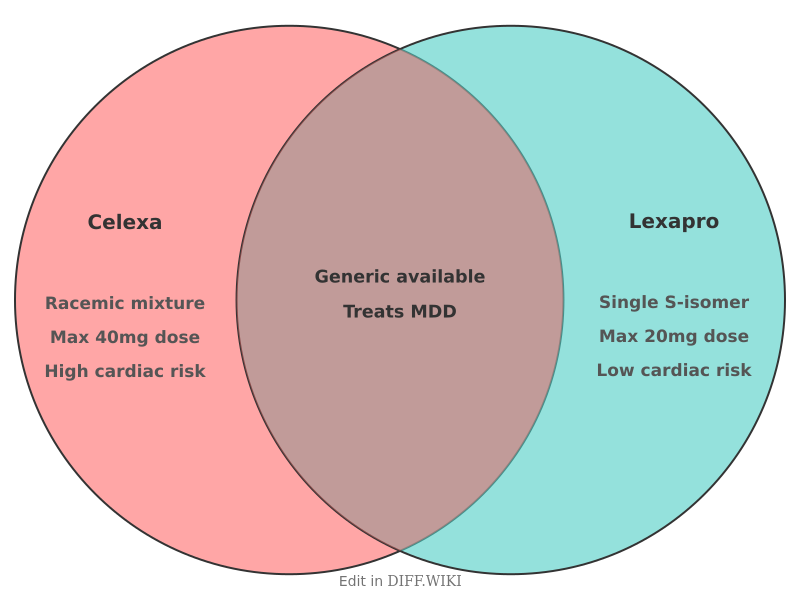
Citalopram (brand name Celexa) and escitalopram (brand name Lexapro) are antidepressant medications belonging to the selective serotonin reuptake inhibitor (SSRI) class. Both drugs are used to treat major depressive disorder (MDD). Escitalopram is the pure S-enantiomer of the racemic mixture found in citalopram, which contains both S- and R-enantiomers in equal proportions.[1] While both medications share a similar mechanism of action by blocking the reuptake of serotonin in the brain, they differ in potency, approved indications, and side-effect profiles.
Chemical structure and mechanism[edit]
Citalopram was approved by the U.S. Food and Drug Administration (FDA) in 1998. It is a racemic mixture, meaning it consists of two molecules that are mirror images of each other: R-citalopram and S-citalopram. Researchers found that the antidepressant effects are primarily derived from the S-enantiomer. The R-enantiomer is considered inactive for therapeutic purposes and may interfere with the binding of the S-enantiomer at the serotonin transporter site.[2]
Escitalopram was approved in 2002 as a single-isomer version of citalopram. By removing the R-enantiomer, manufacturers created a more potent medication. Clinical data suggests that 10 mg of escitalopram is roughly equivalent in efficacy to 20 mg of citalopram.[3]
Comparison table[edit]
| Feature | Citalopram (Celexa) | Escitalopram (Lexapro) |
|---|---|---|
| FDA approval year | 1998 | 2002 |
| Chemical form | Racemic mixture (S- and R-enantiomers) | Single isomer (S-enantiomer) |
| Typical starting dose | 20 mg once daily | 10 mg once daily |
| Maximum daily dose | 40 mg (due to cardiac risks) | 20 mg |
| FDA-approved indications | Major depressive disorder (MDD) | MDD, Generalized anxiety disorder (GAD) |
| Pediatric use | Not FDA-approved for children | Approved for MDD in adolescents (ages 12+) |
| Cardiac risk profile | Higher risk of QT prolongation | Lower risk of QT prolongation |
| Generic availability | Yes | Yes |
Clinical indications and efficacy[edit]
Both drugs are primarily prescribed for depression. However, escitalopram has received FDA approval for the treatment of generalized anxiety disorder (GAD), whereas citalopram is often used off-label for this condition. Meta-analyses comparing the two drugs have indicated that escitalopram may have a faster onset of action and slightly higher rates of treatment response and remission in patients with severe depression.[4]
Side effects and safety[edit]
Common side effects for both medications include nausea, dry mouth, sweating, and sexual dysfunction. In 2011, the FDA issued a safety communication for citalopram, stating that doses above 40 mg per day can cause abnormal changes in the electrical activity of the heart, known as QT interval prolongation. This condition can lead to a fatal heart rhythm called Torsade de Pointes. Consequently, the maximum recommended dose for citalopram is lower than it was at its initial release, particularly for patients over age 60.[5] Escitalopram is associated with a lower risk of QT prolongation at standard therapeutic doses.
References[edit]
- ↑ "Escitalopram Oxalate Monograph". PDR Network. 2024.
- ↑ Sanchez C, Bergqvist PB, Brennum LT, et al. "The clinical relevance of escitalopram's allosteric binding." Psychopharmacology. 2003.
- ↑ "Lexapro (escitalopram oxalate) prescribing information." FDA. 2021.
- ↑ Gorman JM, Korotzer A, Su G. "Efficacy comparison of escitalopram and citalopram in the treatment of major depressive disorder." CNS Spectrums. 2002.
- ↑ "FDA Drug Safety Communication: Abnormal heart rhythms associated with high doses of Celexa (citalopram hydrobromide)." FDA. 2011.