Differences between Percocet and Vicodin
Contents
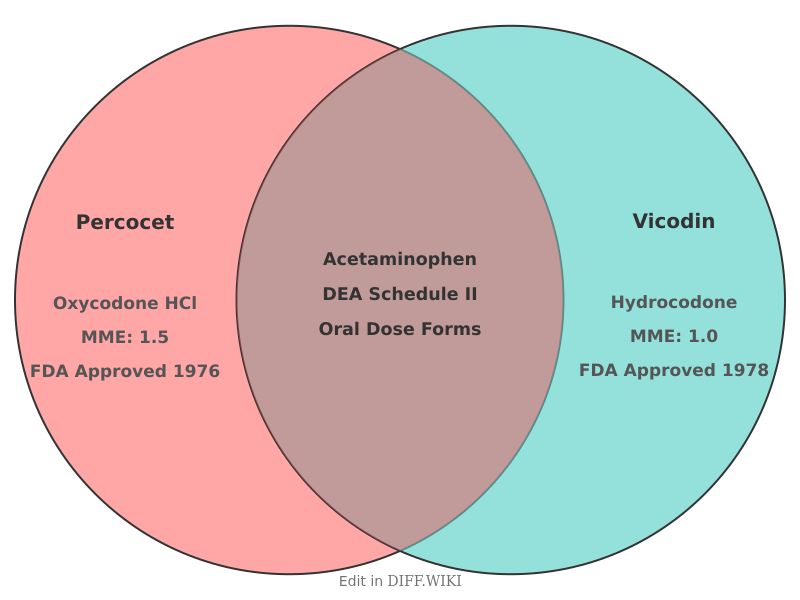
Comparison of Percocet and Vicodin
Percocet and Vicodin are brand-name combination medications prescribed for the management of pain. While both drugs consist of an opioid analgesic paired with acetaminophen, they differ in their primary narcotic component. Percocet contains oxycodone, whereas Vicodin contains hydrocodone. Both medications are classified by the United States Drug Enforcement Administration (DEA) as Schedule II controlled substances due to their potential for abuse, addiction, and severe physical dependence.
Composition and pharmacodynamics
The pharmacological profile of these drugs involves two distinct mechanisms. The opioid component (oxycodone or hydrocodone) binds to mu-opioid receptors in the central nervous system, altering the perception of and emotional response to pain. Acetaminophen, a non-opioid analgesic and antipyretic, inhibits prostaglandin synthesis, which provides additional pain relief and reduces fever.
The Food and Drug Administration (FDA) implemented significant regulatory changes affecting these combination drugs between 2011 and 2014. To reduce the risk of acute liver failure, the FDA mandated that manufacturers limit the amount of acetaminophen in prescription combination products to 325 mg per tablet. Consequently, older formulations of Vicodin that contained 500 mg or 750 mg of acetaminophen were discontinued or reformulated.
Comparison table
| Category | Percocet | Vicodin |
|---|---|---|
| Opioid component | Oxycodone hydrochloride | Hydrocodone bitartrate |
| Non-opioid component | Acetaminophen | Acetaminophen |
| DEA schedule | Schedule II | Schedule II |
| Morphine milligram equivalent (MME) | 1.5 | 1.0 |
| Standard acetaminophen dose | 325 mg | 300 mg to 325 mg |
| FDA approval year | 1976 | 1978 |
| Primary indication | Moderate to severe pain | Moderate to moderately severe pain |
| Common dosage forms | Oral tablet, oral solution | Oral tablet, oral solution |
Clinical efficacy and potency
Research into the relative potency of these medications suggests that oxycodone is approximately 1.5 times more potent than oral morphine. Hydrocodone is generally considered to have a 1:1 potency ratio with oral morphine. A 2005 study published in Academic Emergency Medicine compared the two drugs for the treatment of acute fractures and found no statistically significant difference in pain relief between equivalent doses, although some patients reported higher sedation levels with oxycodone.
The metabolic pathways for these drugs also differ. Oxycodone is primarily metabolized by the CYP3A4 and CYP2D6 enzymes. Hydrocodone is metabolized into hydromorphone via the CYP2D6 pathway. Differences in an individual's genetic expression of these enzymes can influence how effectively they process each medication.
Regulatory history and safety
Historically, Vicodin was classified as a Schedule III substance, which allowed for easier prescribing and refills. However, in 2014, the DEA moved all hydrocodone combination products to Schedule II to address rising rates of diversion and misuse. This change aligned the regulatory status of Vicodin with that of Percocet.
Both medications carry "black box" warnings from the FDA. These warnings highlight the risks of respiratory depression, neonatal opioid withdrawal syndrome, and fatal overdose when combined with alcohol or other central nervous system depressants like benzodiazepines. Long-term use of either drug requires clinical monitoring to mitigate the risk of opioid use disorder.
References
- ↑ Food and Drug Administration. (2014). "FDA statement on Acetaminophen Concentration in Prescription Combination Drug Products." FDA.gov.
- ↑ Drug Enforcement Administration. (2014). "Schedules of Controlled Substances: Rescheduling of Hydrocodone Combination Products From Schedule III to Schedule II." Federal Register, 79(163), 49661-49682.
- ↑ Marco, C. A., et al. (2005). "Comparison of Oxycodone and Hydrocodone for the Treatment of Acute Pain Associated with Fractures." Academic Emergency Medicine, 12(4), 282–288.
- ↑ Dowell, D., et al. (2022). "CDC Clinical Practice Guideline for Prescribing Opioids for Pain — United States, 2022." MMWR Recommendations and Reports, 71(3), 1–95.
- ↑ Sutton, D., & Gossman, W. (2023). "Oxycodone." StatPearls Publishing. NCBI.