Differences between DTaP and Tdap
Contents
DTaP vs. Tdap
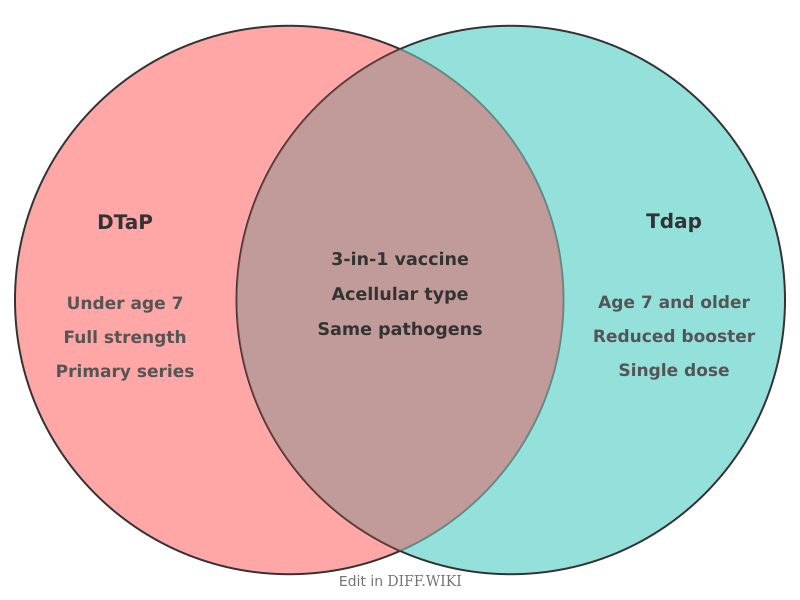
DTaP and Tdap are combination vaccines designed to protect against three bacterial diseases: diphtheria, tetanus, and acellular pertussis. While both vaccines contain the same basic components, they differ in their antigen concentrations and the age groups for which they are approved. In the United States, the Centers for Disease Control and Prevention (CDC) provides specific guidelines on the administration of these vaccines to ensure long-term immunity throughout a person's life.
The uppercase and lowercase letters in the names signify the amount of each antigen included in the dose. An uppercase "D" or "P" indicates a full-strength dose of diphtheria toxoid or acellular pertussis, while a lowercase "d" or "p" indicates a reduced amount used for booster purposes. Both vaccines use an uppercase "T" because the amount of tetanus toxoid is the same in both formulations.
Comparison table
| Category | DTaP | Tdap |
|---|---|---|
| Full name | Diphtheria, Tetanus, and Acellular Pertussis | Tetanus, Diphtheria, and Acellular Pertussis |
| Target age group | Children 6 weeks through 6 years | Adolescents 11 years and older; adults |
| Diphtheria concentration | Full strength (D) | Reduced strength (d) |
| Pertussis concentration | Full strength (P) | Reduced strength (p) |
| Primary purpose | Building initial immunity (Pediatric series) | Maintenance of immunity (Booster) |
| Standard schedule | 5-dose series (2, 4, 6, 15-18 months, and 4-6 years) | One dose at 11-12 years; booster every 10 years |
| Pregnancy indication | Not used | Recommended during every pregnancy (27-36 weeks) |
DTaP (Pediatric formulation)
DTaP is administered to children to help them develop initial protection against the target diseases. The high concentration of diphtheria and pertussis antigens is necessary because the immune systems of infants and young children require a stronger stimulus to create a lasting response. The standard pediatric schedule consists of five doses. If a child misses a dose, healthcare providers use catch-up schedules to ensure the series is completed before the child reaches age seven. DTaP is not licensed for use in individuals aged seven years or older because the higher concentration of antigens is associated with an increased risk of local reactions, such as significant swelling of the arm, in older children and adults.
Tdap (Booster formulation)
Tdap serves as a booster for older children, adolescents, and adults whose immunity from the childhood DTaP series has begun to wane. It was first licensed in 2005 to address the rising numbers of pertussis cases among teenagers and adults. Because the recipient's immune system has already been primed by the initial DTaP series, a lower concentration of diphtheria and pertussis antigens is sufficient to maintain protection.
Medical guidelines recommend that adults who have never received a Tdap dose should get one in place of their next 10-year tetanus (Td) booster. Furthermore, the CDC recommends Tdap for pregnant women during the third trimester of every pregnancy. This timing allows for the transfer of maternal antibodies to the fetus, providing the newborn with temporary protection against pertussis until they are old enough to begin the DTaP series at two months of age.
References
- ↑ Centers for Disease Control and Prevention. (2024). "DTaP (Diphtheria, Tetanus, and Pertussis) Vaccine." Retrieved from https://www.cdc.gov/vaccines/vpd/dtap-tdap-td/hcp/index.html
- ↑ Mayo Clinic Staff. (2023). "Tdap vaccine: Why is it important?" Retrieved from https://www.mayoclinic.org/drugs-supplements/diphtheria-tetanus-and-acellular-pertussis-vaccine-adsorption-intramuscular-route/description/drg-20067342
- ↑ Immunize.org. (2023). "Diphtheria, Tetanus, and Pertussis: Questions and Answers." Retrieved from https://www.immunize.org/catg.d/p4210.pdf
- ↑ U.S. Food and Drug Administration. (2023). "Tdap Vaccines." Retrieved from https://www.fda.gov/vaccines-blood-biologics/vaccines/tdap-vaccines