Differences between Boiling and Simmering
Contents
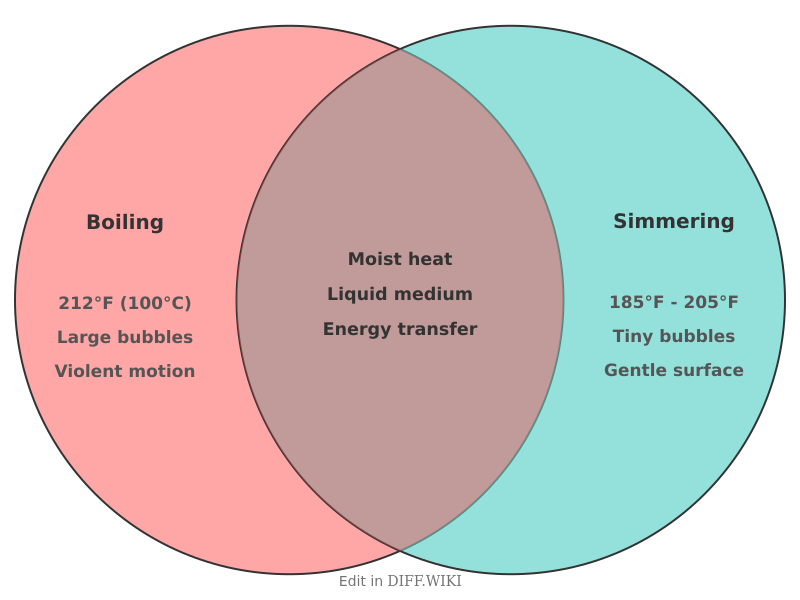
Boiling vs. simmering
Boiling and simmering are moist-heat cooking techniques defined by the temperature of the cooking liquid and the behavior of steam bubbles within that liquid. While both methods involve heating water or a water-based liquid to cook food, the intensity of the heat transfer and the resulting physical agitation of the food differ significantly. Culinary professionals distinguish between these states to control the texture, clarity, and flavor of the final dish.[1]
Comparison table
| Feature | Boiling | Simmering |
|---|---|---|
| Temperature | 100 °C (212 °F) at sea level | 85 °C to 96 °C (185 °F to 205 °F) |
| Bubble Activity | Large, vigorous, and frequent bubbles | Small, gentle, and sparse bubbles |
| Surface Agitation | Violent; the entire surface breaks | Gentle; bubbles barely break the surface |
| Effect on Protein | Toughens muscle fibers; can break delicate fish | Keeps proteins tender; prevents disintegration |
| Liquid Clarity | Clouds stocks by emulsifying fats | Keeps stocks clear by leaving fats undisturbed |
| Common Foods | Pasta, dried grains, tough vegetables | Stews, braises, stocks, and delicate sauces |
Mechanics of boiling
Boiling occurs when a liquid is heated until its vapor pressure equals the atmospheric pressure of the surrounding air. At this stage, steam forms at the bottom of the vessel and rises rapidly, creating large bubbles that agitate the food. This vigorous movement is beneficial for cooking dried pasta, as it prevents the individual pieces from sticking together. The high heat also helps to quickly soften the cellulose in hardy vegetables like corn or carrots.[2]
The boiling point is not constant; it decreases as altitude increases. In high-elevation cities, water boils at lower temperatures, which requires longer cooking times to achieve the same degree of starch gelatinization or protein denaturing found at sea level.
Mechanics of simmering
Simmering is a state of controlled heating just below the boiling point. In a simmer, the bubbles are small—often called "pearls"—and rise slowly from the bottom of the pot. Because the agitation is minimal, simmering is the preferred method for long-cook dishes. For instance, simmering allows the connective tissues in tough cuts of meat, such as collagen, to gradually melt into gelatin without the muscle fibers becoming overly dry or stringy.
In stock making, simmering is essential for aesthetic and culinary quality. A rolling boil will stir together fats and solid impurities with the water, creating a permanent emulsion that makes the stock cloudy. A steady simmer allows these impurities to float to the top, where the cook can remove them with a skimmer.[3]
Sub-boiling variations
Intermediate stages between a cold start and a full boil include poaching and "slow simmering." Poaching occurs at even lower temperatures, roughly 71 °C to 82 °C (160 °F to 180 °F), and is used for extremely delicate items like eggs or fruit. A slow simmer, or "lazy simmer," involves almost no visible bubble movement on the surface and is frequently used for reducing sauces to concentrate their flavors.
References
- ↑ McGee, Harold (2004). On Food and Cooking: The Science and Lore of the Kitchen. Scribner. pp. 782–783. ISBN 978-0684800011.
- ↑ Gisslen, Wayne (2018). Professional Cooking. Wiley. p. 114. ISBN 978-1119399612.
- ↑ The Culinary Institute of America (2011). The Professional Chef. Wiley. p. 264. ISBN 978-0470421352.